|
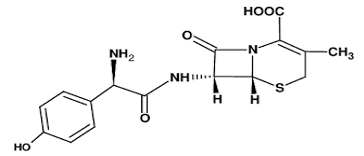
Introduction Cefadroxil is an antibiotic [1] consumed and used to treat mild to moderate infections caused by susceptible microorganisms [2]. It is used to treat bacterial infection of the skin and strep throat for the urinary tract [3,4]. Figure 1 shows the structure of the drug.
Several scientific methods of analysis were available in the literature for the determination of cefadroxil in their pharmaceutical preparations, including fluorimetry [6] and polarography [7,8]. Thin layer chro- matography [9], HPLC [10,11], sequential injection analysis [12], chemiluminescence [13,14], capillary electrophoresis [15], and spectrophotometric methods have been described to determine cefadroxil using various reagents based on the formation of complexes with copper (II) [16] Flow injection analysis (FIA) [17]. Additionally, another method is based on the liberation of hydrogen sulfide and followed by the reaction with N,N-diethyl-p-phenylenediamine [18]. Other spectrophotometric methods are reported for the determination of cefadroxil based on its reactivity with iodine [19]. Nitrosation and subsequent metal chelation reaction with 2,6-dichloro-quinone-4-amino-antipyrene in the presence of potassium hexacyanoferrate [20] or by oxidation in an acid medium [21]. These methods are time-consuming and required extraction steps or required indirect procedures. This work describes a simple and sensitive spectrophotometric method for the determination of cefadroxil. This method is based on the reaction of the drug with NQS and the formation of Schiff's base.
Experimental Apparatus
A meter electrical balance was used to weigh the sample. The reagent was supplied by BOH and Fluka. The standard solution of 100 ppm cefadroxil was prepared by dissolving 0.01 g in 2 ml of de-ionized water and then diluted to 100 ml. Also, 5 × 10-3 M of 1,2-naphthoquinone reagent was made by dissolving 0.06 g in 50 ml de-ionized water.
Results and Discussion In a primary test, the NQS reagent reacted with cefadroxil in the presence of sodium hydroxide NaOH and formed a red color product with the highest absorption peak at 465 nm, where the reagent blank showed low absorbance at this wavelength.
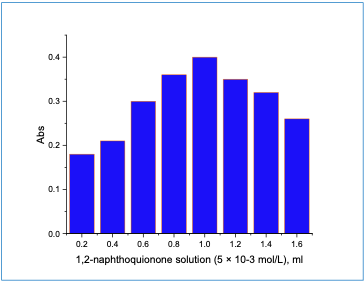
Study of Optimal Reaction Conditions Impact of NQS Reagent The impact of changing the reagent 1,2-naphthoquinone solution concentration onthe absorbance of cefadroxil was performed. It was noticed that the absorbance increasedand reached a maximum when 1 ml of 5 ×10-3 M 1,2 - naphthoquinone solution wasused (Figure 2).
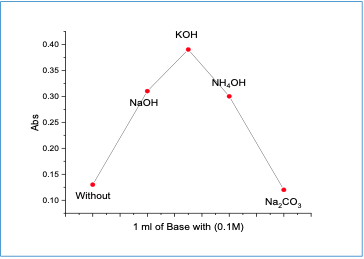
Impact of Base The impact of bases (sodium hydroxideNaOH, potassium hydroxide KOH,ammonium hydroxide NH4OH, and sodiumcarbonate Na2CO3) was investigated. It wasfound that potassium hydroxide gave maximum absorption at 460 nm (Figure 3).
Furthermore, the impact of the potassium hydroxide volume and pH were studied. Maximum absorbance was observed when 1.5 ml of 0.1M KOH at pH 11.31 was used.
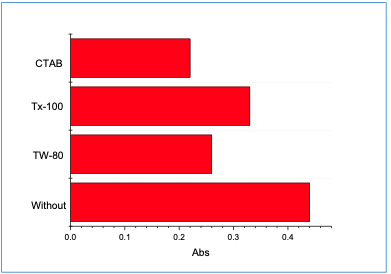
Impact of Surfactants The impact of Tween 80 (TW-80), Triton X- 100 (TX-100), and cetyltrimethyl ammonium bromide (CTAB) of 0.1% concentration was studied. However, the absorbance was decreased when CTAB was used (Figure 4). Therefore, it was excluded from the experiment.
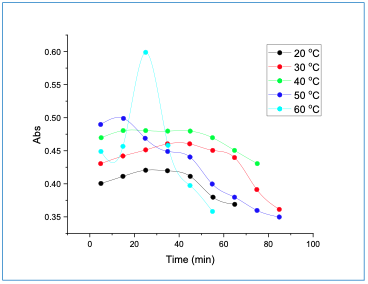
Impact of Temperature Versus Time on the Absorbance of the Complex The effect of reaction time was performed at different temperatures. Figure 5 shows a decrease in the absorbance when time increased, attributed to the dissociation of the complex. It was found that the optimum time and temperature for the complex was 15 min at 40°C, respectively.
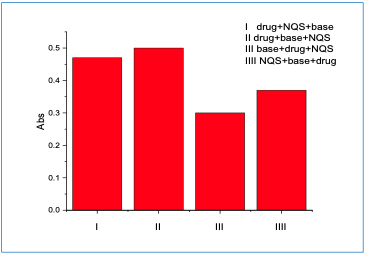
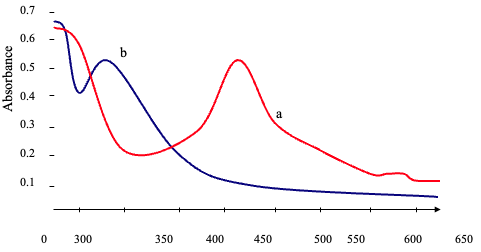
Impact of the Order of Addition Under the optimum conditions, the order of addition was investigated. Figure 6 shows that in the order of addition, no. II was the best. Absorption Spectra Figure 7 shows the absorption spectra for the best condition that has been confirmed above.
The Details of the Statistical Data and Optical Characteristics of the Suggested Method The absorbance of the complex was measured at 460 nm. Beer's law limits and molar absorptivity values are shown in Table 2. In addition, the relative standard deviation (RSD) and the accuracy of analysis on six replicates for three different concentrations of cefadroxil indicate that the method is valid. Also, the limit of detection (LOD) is accepted as well.
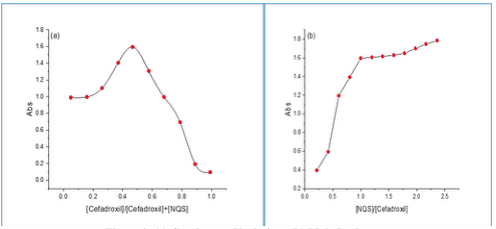
Analytical Implementation The results showed that the experimental F-Test and T-Test were less than the theoretical value (t = 2.50, f = 6.41). However, it was observed that there was no significant variation between the suggested method and the formal method [22]. Quantities and Stability Constant Quantities of a reaction of cefadroxil for NQS were studied through the molar ratio as well as job method [23,24].
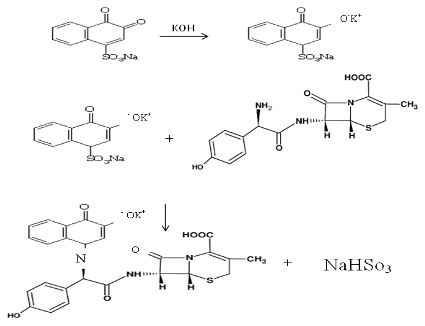
Figure 8 (a) and (b) showed that the results were 1:1 and the average conditional stability constant for the resulting complex was calculated using the equation (1) below: where Kst: the stability constant (L/mol), (∝): the dissociation degree, and (C): the concentration of the resulting complex. The average Kst is 2.7 × 106 which illustrates that the resulting product is stable. Mechanism of the Reaction Under the experimental conditions, the mechanism of the reaction is shown in Scheme 1.
The mechanism suggests that the NQS was converted into a quinoidal which reacts with phenol amine via the replacement of the hydrogen atom of the primary aromatic amine group to produce paraquinoidimide- condensation (Schiff's base) NaHSO3.
Conclusion The suggested spectrophotometric method is simple, sensitive, and low cost. In addition, this method does not involve a solvent extraction step. Also, it gives accurate and precise results. The calibration curve shows high linearity. The coefficient correlation was higher than 0.99. The limit of detection and limit of quantitation values were very acceptable as well. Finally, the suggested mechanism of the product formation shows the NQS was converted to a quinoidal that reacts with phenol amine to produce paraquinoidimide-condensation (Schiff's base) and NaHSO3.
References
|
||||||||||||||||||||||||||||||||||||||||