|
Introduction Over the past two centuries, extensive investigations into the chemical and biological activities of plants have catalyzed advancements in synthetic organic chemistry, giving rise to the field of medicinal chemistry. This evolution has paved the way for the development of more potent therapeutic agents [1]. As observed in recent years, multiple drug resistance in human pathogenic microorganisms have developed due to indiscriminate use of commercial antimicrobial drugs commonly used in the treatment of infectious diseases. This situation forced scientists to search for new antimicrobial substances from various sources, like medicinal plants, which are good sources of novel antimicrobial chemotherapeutic agents [2]. Medicinal plants have been used for centuries as remedies for human diseases because of their biochemical constituents, which have therapeutic significance [3]; also because of their ability to biosynthesize a diverse array of chemical compounds with profound physiological significance. They are harnessed for human use across various domains including perfumery, culinary arts, cosmetics, and pharmaceuticals. These plants can synthesize a large variety of chemical substances that are of physiological importance [1,4]. This paper is concerned with studying the plant Juniperus, classified as a conifer plant within the Cupressaceae family. It is estimated that 70 species of Juniperus are distributed throughout the world, primarily North America, Europe, and Asia [5,6]. Juniperus represents the third largest genus among global conifers, characterized by its monophyletic nature. Remarkably long-lived, some Juniperus specimens have demonstrated a lifespan of up to 2000 years and have shown a variety of sizes from small shrubs to towering forest giants [6,7]. Notably, Juniperus exhibits a unique resilience to extreme temperature fluctuations, enabling its growth in arid environments that prove inhospitable to many other flora [8]. In addition, the wood derived from Juniperus is distinguished by its aromatic qualities and its resistance to decay. This unique combination of attributes has made it highly sought after for a variety of applications, including the manufacture of furniture, panels and barriers. Conifers such as Juniperus are an excellent source of fire- wood, and tend to produce minimal ash and smoke when burned [6]. Moreover, the genus Juniperus has earned recognition for its prominent role in traditional medicine [9]. J. excelsa: this medicinal plant has been historically employed in the treatment of various conditions including dysmenorrheal, cough, bronchitis and colds, jaundice and tuberculosis, and to induce menses and expel fetuses [2,5], abdominal spasm, asthma, diarrhea, fever, headache, gonorrhea, and leucorrhoea. It is also considered useful as an antihypertensive, appetizer, diuretic, carminative, and stimulant [10]. Juniperus excelsa M.Bieb. is a large shrub or tree, spread mainly throughout the eastern Mediterranean, starting from north-eastern Greece and southern Bulgaria, across Turkey, to the Middle East countries (Syria and Lebanon) and the Caucasus Mountains. Its presence is also noted in Iran, Pakistan and Oman [11]. J. excelsa is rich in phenolic compounds and demonstrates various biological effects, such as antimicrobial, antifungal, antioxidant, anti-inflammatory, anticancer, antiviral, and cytotoxic activities. Phytochemical analyses have unveiled the presence of sterols, flavonoids, lignans, polysaccharides, some aromatic compounds, and fatty acids across various anatomical components of the Juniperus genus [12]. Since antioxidants were discussed, it must be mentioned that it has recently been observed that reliance on synthetic antioxidants in food production leads to a deterioration of the taste and biologically active qualities of food and may lead to conditions suitable for the occurrence of food allergies. This has spurred researchers to seek alternative sources of reactive components capable of counteracting oxidative processes, which have negative effects on cellular metabolism. Knowledge of the antioxidant properties of many plant species allows their usage as a means of preserving food quality by slowing down or preventing lipid oxidation processes. The pursuit of novel and safe antioxidants from natural sources has garnered substantial interest in their application for natural antioxidants, functional foods, and nutraceuticals [12-14]. Based on the above, the goals of the present study were to assess the antimicrobial potential of aqueous and ethanolic extracts Juniperus excelsa M.Bieb.against Gram-positive bacteria, Gram-negative bacteria, and yeast; to evaluate the antioxidant activity of plant extracts; and to determine the total phenolics content of plant extracts and perform the phytochemical screening of dried leaves of the plant studied.
Materials and Methods The study was conducted in the laboratories of Syrian Private University, Atomic Energy Commission of Syria, and the National Commission for Biotechnology in Damascus. Plant sample The leaves of Juniperus excelsa M.Bieb. were collected in July 2023 from the al- Khusha in the mountains of al-Qalamoun, Ras al-Ma'arra village, Yabroud area, Damascus countryside, Syria. The fresh leaves were separated from the bark and then washed with water thoroughly and dried for 24 hours at room temperature in shadow. The leaves were stored at -20°C until use, then crushed immediately before using into small pieces using an electric grinder, where the particle dimensions were less or equal to 0.2 mm. Figure 1 shows Juniperus excelsa M.Bieb. trees with the taxonomy of the plant [15].
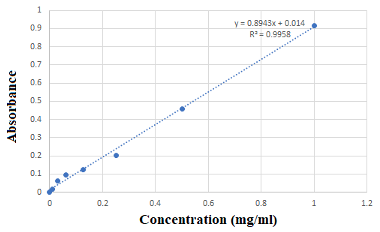
Proximate Composition Analysis The proximate analysis of dried leaves of Juniperus excelsa M.Bieb. was carried out for moisture content, protein, fat, carbohydrate, crude fiber and ash content as described by the Association of Official Analytical Chemists (AOAC) (2000). All chemical analysis results were calculated relative to the oven dry sample weight [16]. Mineral Composition Analysis Dried leaves of Juniperus excelsa M.Bieb. were mineralized at 450°C. The residue was first dissolved in concentrated HCl and evaporated to dryness, then the remainder was dissolved in 0.1 mol/L HNO3 solution. Mineral contents were determined on an atomic absorption spectrophotometer (AAS) Perkin Elmer/ HGA 500 (Norwalk, USA), under the following instrumental parameters for the flame AAS: sodium (Na) 589.6 nm; potassium (K), 766.5 nm; calcium (Ca), 317.0 nm; and iron (Fe), 238.3 nm. Identification of metals was carried out by comparison to a standard solution of metal salts, and metal concentrations were calculated from a calibration curve, built by using a standard ppm solution [17]. Preparation of Plant Extracts 50 g of dried Juniperus excelsa M.Bieb. leaf powder was extracted with 200 mL of water or ethanol with continuous stirring by a magnetic stirrer at 25°C and 6 rpm for 6 hours; after which, each mixture was soaked for 24 hours at 4°C and then was filtered using Whitman No. 1 filter paper. Each supernatant was evaporated using a vacuum rotary evaporator to concentrate the extracts. The volume of the resulting aqueous extract was 3.5 mL, while the volume of the ethanolic extract was 3 mL. Total content of phenolic compounds Phenolics are secondary metabolites that are omnipresent in plants. They are a large group of bioactive compounds comprising about 8000 compounds [18]. The two major classes of phenolic compounds include flav- onoids and phenolic acids [19]. The total phenolics of Juniperus excelsa M.Bieb. extracts were determined by the Folin–Ciocalteu method with some modifications commensurate with laboratory work. 0.0025 mL of each extract was mixed with 0.5 mL of Folin–Ciocalteu reagent in complete darkness for 10 min, and then 1 mL of sodium carbonate solution 15% was added. The extracts were kept in the dark for 30 min and the absorbance was measured at 740 nm. A calibration curve with gallic acid (1 mg/mL) was established, where (0.01, 0.03, 0.06, 0.125, 0.25, 0.5, 1) mg/mL of gallic acid solution were used instead of the studied sample, and the total phenolic content was expressed in milligrams of gallic acid per mL of Plant extract. Figure 2 shows the standard curve of gallic acid [20,21].
Determination of DPPH-Free Radical Scavenging Ability The antioxidant activity of extract samples was assessed on the basis of radical scavenging effect of the stable DPPH (1,1- diphenyl-2-picrylhydrazyl) free radical. The DPPH• test is based on the ability of the stable 2,2-diphenyl-1-picrylhydrazyl free radical to react with hydrogen donors. The DPPH• radical displays an intense UV-VIS absorption spectrum. In this test, a solution of radical is decolourized after reduction with an antioxidant (AH) or a radical (R•) in accordance with the following scheme: This method is very simple and quick for manual analysis [20]. In a test tube, 1 mL of ethanolic DPPH solution (4 mg/100 mL absolute ethanol) was combined with 50 μL of each extract. After that, the tubes were kept in darkness for 30 min. The absorbance was therefore determined at 517 nm. The scavenging rate of a DPPH-free radical was calculated according to the following formula:
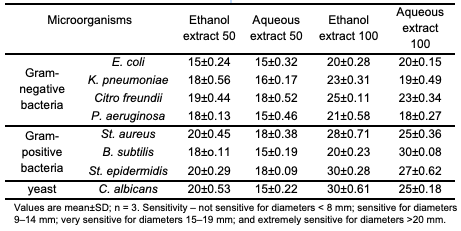
where, AControl is the absorbance of the control reaction (containing all reagents except the test compound), and ASample is the absorbance of the test compound [23]. Antimicrobial activity Screening of the antimicrobials was investigated on Gram-negative bacteria (E. coli, K. pneumoniae, Citro. freundii, P. aeruginosa), Gram-positive bacteria (St. aureus, B. subtilis, St. epidermidis) and yeast (C. albicans). The disc diffusion method was employed to determine the antimicrobial activities of the studied extracts. The parent cultures of each microorganism were set up to 24 hours before the assays to reach the stationary phase of growth. Briefly, a suspension of the tested microorganism that contained 106 colony-forming unit/mL was prepared and then spread on a solid medium (nutrient agar) by a swab. Paper disks (diameter 6 mm) were impregnated with different amounts of each extract (50 and 100 μL), placed on inoculated plates and left for 15 minutes at room temperature. The plates were incubated at 37°C for 24 hours for bacteria and at 27°C for 48 hours for the yeasts. The diameters of inhibition zones were measured in millimeters [5]. The sensitivity to the extracts was determined by the diameter of the inhibition zones: not sensitive for diameters <8 mm; sensitive for diameters 9– 14 mm; very sensitive for diameters 15–19 mm: and extremely sensitive for diameters >20 mm [10].
Statistical Analysis Statistical analysis was carried out using the STATISTICA 12.0 version (StatSoft, Inc., USA). Each experiment was conducted 3 times; all data were presented as mean values with their standard deviations (mean±SD). The analysis of variance (ANOVA) was performed, with a confidence interval of 95% (P < 0.05). Means were compared by Duncan multiple range test.
Results and Discussion Knowledge of proximate, micronutrients and phytochemical composition is funda- mental to the understanding of modes and mechanisms of action of medicinal plants in general and play a crucial role in assessing its nutritional value and significance (<0.05) [24]. The main chemical, nutritional and mineral composition of J. excelsa M.Bieb. dried leaves was determined, and the data pertaining to various chemical and mineral composition are illustrated in Table 1 and Table 2.
Results given in Table 1 indicated that the plant is a rich source of basic food nutrients such as proteins, fats, fibre, and carbo- hydrates. The values of these substances amounted to 5.7%, 10.6 %, 49.1%, and 22.2%, respectively, while the moisture and ash contents were 7.94% and 10.6%, respectively. All these substances have an important place in human nutrition. Dietary fibres are constituents of many fruits and vegetables. Clinical studies suggest fiber can exert a wide range of benefits in areas such as bowel function, gut health, immunity, blood glucose control, and serum lipid levels [25]. On the other hand, high fibre content for the leaves of the plant is usually a major drawback in human nutrition as it can cause intestinal irritation and it is low in nutrient availability as humans cannot digest them easily [26]. Additionally, dietary fibre delays gastric emptying or increases viscosity of GIT content thereby suppressing digestion and carbohydrate absorption. This mechanism is selectively advantageous in that the threat or risk of hypoglycaemia, hyperinsulinemia and undue weight-gain is absolved [24]. Carbohydrates are useful as they supply energy to cells such as brain, muscles and blood. They contribute to fat metabolism and spare proteins as an energy source and act as a mild laxative for human beings. They generally add to the bulk of the diet [26,27]. The protein content of the leaves shows the plant is a good protein supplement for animal feed [26], however, the content is not as high as the 12.5% reported for Nauclea latifolia leaves, 20.42% for Moringa leaves and 14.73% for Ageratum conyzoides leaves [25-27]. The fat content (10.6% for the leaves of J. excelsa M.Bieb.) is low and compares with the results for Agbafor et al. (2015) and Eze et al. (2014), who concluded that the leaves of Ageratum conyzoides are poor sources of lipids [25,26]. Our results agree with Dike et al. (2016) [28]. The total moisture content is considered low, and this low moisture content of the leaves indicates that the plant materials can be stored over a long period with lesser susceptibility to microbial attack [28]. The total ash content of the plant materials is low (4.5%) indicating low total mineral elements in the plant materials [29]. Minerals have an important place in human nutrition. The importance of these minerals, also known as trace elements, has been well understood in recent years, since when missed, causes diseases and major disorders in the human body [30]. The mineral composition of J. excelsa M.Bieb.dried leaves was analyzed and the results revealed that iron was 0.1, calcium 2500, potassium 830, and sodium 125 ppm. These minerals play significant roles in several biological processes. Bone growth and turnover are influenced and regulated by the metabolism of Ca and other minerals, in addition to its role in muscle contraction and blood clotting. Iron is an essential trace element for normal functioning of the central nervous system. It is known to be a component of some metalloenzymes, myoglobin and hemoglobin, which is needed in the transport of oxygen and carbon dioxide during respiration or cellular metabolism. This hemoglobin (containing iron) also serves as a buffer to regulate changes in blood. Potassium and sodium, which are presented in the intracellular and extracellular fluid, help to maintain electrolyte balance and membrane fluidity pH [29]. Also, potassium is involved in inducement of calmness [25]. The amount of Ca was close to its content in Momordica charantia leaves while the amount of Na, K, and Fe was lower [29]. The balanced nutritional composition of J. excelsa M.Bieb.makes it a potential source of nutrients that can be included in a number of foods. Total Content of Phenolic Compounds The amounts of the total polyphenols in the aqueous and ethanol extracts of J. excelsa M.Bieb.leaves are shown in Table 3. The total phenolic content found in water and ethanol extracts was 0.631 and 1.27 mg/mL, respectively. The values were reported as mg of gallic acid equivalent (GAE) per 1 mL of extract with reference to gallic acid standard curve.
Results given in Table 3 indicate that the ethanol extract shows high total phenol content. The result is similar to those reported by Rebaya et al. (2014) and Do et al. (2014) [13, 31]. Phenolics are the most predominant phytochemicals present in nature, and to date, about 10,000 different structures are currently described [32]. Phenolic components represent the largest class of secondary metabolites which are known as exceptional antioxidants, and their antioxidant activity is based on their redox potential. They include phenolic acids, flavonoids, tannins, stilbenes, curcuminoids, coumarins, lignans, quinones and others. Various bioactivities of phenolic compounds are responsible for their biological properties, like antimicrobial, anticarcinogenic, anti-inflammatory and therapeutic effects [33], and also play a major role in modifying gene expression [18]. Phenolic substances have important contributions to human health. These compounds have been reported to play an effective role in preventing cancer and cardiovascular diseases by preventing free radical formation in the body [30]. The presence of phenolic compounds enables the plants to act as reducing agents, hydrogen donators and singlet oxygen quenchers. During cell metabolism, all living cells generate free radicals as part of normal cellular functions that are highly reactive may cause oxidative stress, leading to cellular damage and subsequently various diseases, such as atherosclerosis, diabetes mellitus, arthritis, ischemic heart disease, gastritis, immunosuppression, neurodegenerative diseases, ageing, and cancer [19]. DPPH Radical Scavenging Activity Determining antioxidant activity as radical scavenging activities are critically important due to the detrimental role of free radicals in foods and biological systems. Plants rich in secondary metabolites such as phenolics and flavonoids exhibit antioxidant activity attributable to their oxidative properties and chemical compositions. The DPPH assay is one of the indispensable tests when assessing the antioxidant activity of extracts and is widely used to test the free radical-scavenging activity of several natural products. The results of the antioxidants for aqueous and alcoholic extracts of J. excelsa were demonstrated in Table 4.
DPPH reacted, as a stable free radical, interacting with any molecule capable of donating an electron or hydrogen to it, which lead to DPPH bleaching (color change from violet to yellow). The absorbance was measured at 517 nm [34]. The data presented in this study supports the prevailing mechanism which designates DPPH• as a primary, if not exclusive, hydrogen acceptor [35]. The enhanced DPPH scavenging activity observed may be attributed to higher phenolic contents [34]. The results also indicate that the ethanolic extract exhibits greater activity compared to the aqueous extract. That is due to the phenolic compounds' major contribution to antioxidant properties of the used plant. This may be explained by the fact that different types of phenolic compounds possess dif- ferent antioxidant capacities which is related to their chemical structure. The results of this study are in agreement with the results of many studies that reported that the plant extracts with high total phenolic contentshowed high radical scavenging activity, formed from very complex mixtures of different molecules, and the proton donating hydroxyl groups with a particular position in the structure of molecules might be controlling the radical scavenging properties of these extracts [36]. The results could be ex- plained by the sensitivity of the Folin- Ciocalteu reagent towards a wide range of phenolic compounds, whereas DPPH free radicals display varying sensitivity to different antioxidants. The Folin-Ciocalteu reagent reacts with both free and bound phenolics present in the extracts and other samples, whereas the DPPH assay exclu- sively quantifies free antioxidants and phenolics [14]. The existing literature provides evidence of a significant correlation between total phenolic content and antioxidant activity, suggesting that phenolic compounds make a substantial contribution to the antioxidant properties of the respective plant materials [36]. Antimicrobial Activity Assay In the present study, the antimicrobial compounds from the leaves of J. excelsa M.Bieb. were extracted against wide range of microorganisms on the basis of disc-diffusion assay using a suspension containing contained 106 colony-forming unit/ mL microorganism spread on nutrient agar. The discs (6 mm in diameter) were impregnated with 50 and 100 μL separately of each extract and placed on the inoculated agar. The antimicrobial activities of J. excelsa M.Bieb.ethanol and aqueous extracts against microorganisms were examined in the present study, and their potency was quantitatively assessed by the presence or absence of inhibition zones and zone diameters. The inhibition zone, measured in millimeters, including the diameter of the well, was used as the criterion for measuring the antimicrobial activity. The results are as shown in Table 5 showed that the ethanol and aqueous extracts have an inhibition effect on the growth of all microorganisms tested with varying degrees of inhibition, depending on the bacterial strains and volume of extract applied.
The diameter of inhibition zones was obtained in the range of 15 to 30 mm. The ethanol extracts of the leaves showed the highest activity on all microorganisms, except B. subtilis at 100 μL, probably because of more efficient extraction by ethanol. Behbahani et al. (2013) reported that alcoholic extract of Eucalyptus camaldulensis leaves has a greater impact on all strains compared to aqueous extract [37]. Also, extracts (100 μL) showed higher activity than extracts (50 μL). For Gram-positive, the most sensitive microorganisms were St. aureus, St. epidermidis with 20 mm inhibition for ethanol extract (50 μL), and St. epidermidis with 30 mm inhibition for ethanol extract (100 μL), St. aureus and St. epidermidis with 18 mm inhibition for aqueous extract (50 μL), and B. subtilis with 30 mm for aqueous extract (100 μL). On the other hand, the most sensitive Gram-negative bacteria was Citro. freundii at all extracts with the range 18-33 mm inhibition. This bacterial inhibition is mainly related to the chemical composition of the extracts [4]. These kinds of differences in susceptibility among the microorganisms against antimicrobial substances in the plant extracts may be explained by the differences in cell wall composition and/or inheritance genes on plasmids that can be easily transferred among bacterial strains [2]. Our data indicate that Gram-positive bacteria are the most sensitive tested strain to the different extracts; the tendency of polyphenols, flavonoids, tannins, and anthocyanins to react could be explained by that. The structures of the cell envelope, including cytoplasmic membrane and cell-wall component, are different between Gram-positive and Gram-negative bacteria. Gram-negative bacteria possess an outer membrane surrounding the cell wall, which restricts diffusion of hydrophobic compounds through its lipopolysaccharide covering. Without the outer membrane, the cell wall of Gram-positive bacteria can be permeated more easily and disturb the cytoplasmic membrane, disrupting the proton motive force, electron flow, active transport, and coagulation of cell contents. Therefore, the structural difference of bacteria plays an important role in their susceptibility [9]. In many studies, the mechanism of the cell wall is considered. Kotzekidou et al. (2008) find that the antimicrobial compounds in the plant extract have interaction with the phospholipids’ two layers membrane, and affect the permeability of the bacterial cell membrane, and released the intracellular components. Also, they have reported that cell wall and cell membrane affected and changed their permeability, causing the release of intracellular contents, which can be ass- ociated with impaired membrane function, such as electron transfer, enzyme activity or nutrient uptake [37,38]. In conclusion, it can be suggested that J. excelsa M.Bieb. leaves extract in vitro have considerable antimicrobial ability over the studied strains. More studies are needed to identify the effective dose of the extract on the micro- organisms, and introduce the extract as a natural and novel antimicrobial compound. Therefore, using J. excelsa M.Bieb. as a natural antimicrobial compound in vitro requires further research on the mechanism of the pharmacy plant on microorganisms.
Conclusion Medicinal plants are useful and economically essential. J. excelsa M.Bieb. is a plant of medicinal importance due to its activity against microorganism. The extracts obtained by J. excelsa M.Bieb. plant showed significant antimicrobial and antioxidant activities, which can be used as anti-microbial agents in new drugs for therapy to treat many infectious diseases. Two extracts were obtained, i.e. ethanolic extract and aqueous extract from J. excelsa M.Bieb. leaves. The quantitative analysis of J. excelsa M.Bieb. showed that the phenol content was 1.27 and 0.631 mg/mL and antioxidant activity was 65.13 and 27.63% for ethanolic and aqueous extract, respectively.
Author Contributions Investigation, Project administration, Supervision, and Software; D, Mariam. Writing–review & editing, Formal analysis, and Methodology; A. H, Manar. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding. Data Availability Statement: Data is contained within the article. Conflicts of Interest: The authors declare no conflict of interest.
References
|
||||||||||||||||||||||||||||||||