|
Introduction Plant based medicines (Aayurveda) are safer alternatives to many synthetic medicines. They have fewer side effects, and the body can absorb these bio-compounds easily [1- 4]. Even though this natural medicine is considered to have many advantages, the main drawback is that it needs more curing time, and larger amounts of the plants are essential for the drug’s preparation [5-9]. The main alternate solution for such problems is to concentrate the particular components of the plant which cures and to synthesize the same chemically [10-11]. So, we may have to consider the whole plant extract and it’s chemical components, which are actually responsible for healing [12]. While choosing the synthesis of the same chemically, we can minimize its excess use and presence of other components in the plant which may cause complications [13]. That is, we may suggest the medicinal com- pound in pure form through chemical synthesis without destroying the natural sources. Plectranthus zeylanicus (PZ) is a perennial herb, cultivated in Sri Lanka and in India. This plant is known as karpuravalli and iruveli in Malayalam and kannikkaurkka in Sanskrit. Because of the presence of bioactive secondary metabolites, PZ is used in many traditional drugs [14-16]. The plants as a whole or as various extracts are used for the treatment of dysentery, eye disorders, vomiting, dermatitis, throat in- fection, cough, fever, and ulcers. The major component of this plant is α-terpeneol [17]. More than 100 compounds of this plant were detected, presented in small amounts. That is, only 60% of the components were identified. P-cymene, geraniol and geranyl acetate were found. 6β, 7β- dihydroxy-royleanone were also identified. It contains aromatic chemical compounds and essential oils, and has anti-inflammatory, antifungal, antimicrobial and disinfectant activities. These plants are very important for the perfume and medical industry [18]. Mayuri Napagoda et al. [19] have isolated dichloromethane extract of PZ and obtained the compound 7α-acetoxy-6β-hydroxy-royleanone. In another study, Mayuri Napagoda et al. [21] revealed 5 lipoxy- genase(5-Lo) inhibition of PZ, which has an important role in preventing disorders like asthma or atherosclerosis. They have studied the anti-inflammatory activity of the isolated phytoconstituent in-vitro. They also studied its antibacterial and antifungal activities and claimed this as a remedy for inflammatory disorders and microbial infections. Sudhara G. De Soyaza et al. [20] studied the antimicrobial activity of silver nanoparticle prepared from PZ. They have studied the hexane extract of this plant and indicate the potent inhibition of 5-lipoxygenase enzyme. They have also claimed the presence of diterpenoids, pentacyclic triterpenes, fatty acids, phytosterols, etc., in the extract. In this paper, we describe the extraction of PZ using ethanol solution. The green plants planted in our farm were collected, washed and dried in shade [21-23]. The plant was identified by a botanical expert. They were subjected to polarity index extractions, using organic solvents. For the specific study, we collected the ethanol extract. Our aim was to extract the phytochemical components, separate and purify and reproduce them in high concentration and use them in the affected areas [24-26].
Experimental Materials Plectranthus zeylancius (Figure 1), Hexane, Ethyl Acetate (E Merck, India), Ethanol (commercial grade). All solvents were distilled before use.
Instruments UV spectra were recorded on an Ultravioletvisible spectrophotometer (Jasco V-650), FT-IR spectrum was produced by Fourier transform infrared spectrometer (FT-IR) (Jasco model 4100) using KBr pellets. Liquid Chromatography Mass Spectrum was obtained by Agilent 6100 Series Quad- rupole. The NMR spectral study was carried out at 400 (1H) and 100 (13C) MHz on a Bruker-400 MHz spectrometer instruments. The chemical shift values are reported relative to Me4Si (1H) and CDCl3 (13C) as internal standards. The value of coupling constant (J) was stated in Hertz (Hz).Procedure The dried and powdered sample was used for the analysis. It was extracted using solvents of increasing polarity. First, hexane was used. The components soluble in hexane were separated out. The second extraction was carried out with ethyl acetate and the compounds were separated. In the third step, we selected ethanol as a solvent. 30 g of the powder of stem and leaf of PZ was transferred to the round bottom flask and extracted using ethanol as a solvent for a time span of three hours. It was filtered and the ethanol extract was collected. The solvent was removed. The dry army green coloured compound, thus obtained, was subjected to TLC and then purified by col- umn chromatography. A single component was obtained (Figure 2) and used for further study.
The structural elucidation was carried out using UV, IR, NMRs and LC-MS spectra and information from literature.
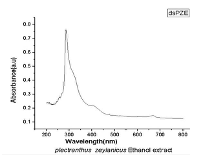
Results and Discussion As a whole, we have obtained a very low yield of 800 mg pure compound from 30 g of starting material. For the structure elucidation of the compound, the following spectra were recorded.Spectral Details Figure 3 shows the UV-visible spectrum of ethanol extract of PZ. It shows three peaks at 286, 310 and 664 nm. The first peak (286 nm) indicates the presence of unsaturation (𝛑-𝛑* transitions) and the second peak (310 nm) for a carbonyl group (n- 𝛑*transitions). The peak obtained in the visible region (664 nm) is of its colour.
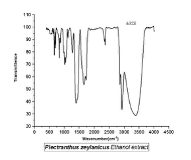
Figure 4 shows the IR spectrum of ethanolextract of PZ. The peaks (cm-1) obtained are at 3557 (OH), 3345 (C-H str of C=C), 2918(C-H str of CH3), 2849 (C-H str of CH),1716 (C=O), 1644 (C=C bend), 1603 (C-N),1383 (C-H bend), 1262 (C-O aromatic),1116 (C-Oaliphatic).
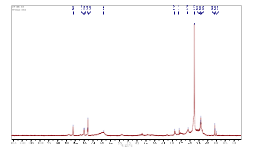
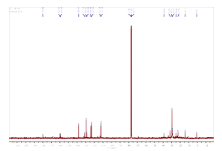
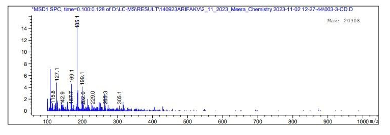
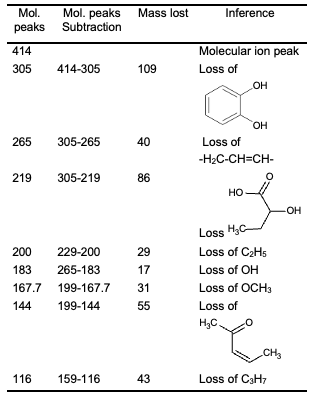
Figures 5 and 6 give the NMR spectra of the compound. The1H- NMR spectrum of the compound shows the peaks (ppm) at 8.10 (s) (CHO), 7.48 (d), (ar H), 7.26 (solvent CDCl3) 6.37 (s) (CH=C), 2.36 (s), 2.11 (s), 1.61 (s), 1.25 (s), 0.89 (t), 0.08 (t) (aliphatic hydrogen). From 13C-NMR spectroscopy, we obtained peaks (ppm) at 170.98 (C=O), 150.5, 130.4, 128.8, 123.2 (ar C), 112.2 (C=C), 36.6, 31.9, 29.37, 24.66, 22.7, 14.5 (aliphatic C). Figure 7 shows LC-MS spectrum of the ethanol extract of the PZ. The maximum molar weight obtained is 305. The most abundant peak is observed at 185. The fragmentation pattern is given in Table 1.
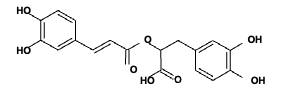
As per the UV spectrum, we have a C=O and unsaturation in the molecule. From IR spectrum, we have C=C, CH3, CH, C=O, C- N, OH and C-O groups. The mass spectrum gives the possibility of groups that are shown in Table 1. From the literature values of various extracts of PZ, the following structure (Figure 8) is almost suited for our extract.
The positions of the substituents are maintained at the same place. The phenolic groups may be substituted with alkyl chains. As per the mass spectrum, the molar mass is 414 (M+ peak) and the proposed structure has the molar mass of 416.
Conclusion Phytomedicines are more important in curing various chronic diseases. Plant bodies contain many chemical components in different ratios. When we analyze the therapeutic application of phytochemicals, the structural details are necessary. In this paper, the extraction of Plectranthus zeylancius using ethanol has been carried out. Spectroscopic techniques such as UV, IR, NMR and LCMS were used for structural elucidation. The peaks in the UV- visible spectrum show the presence of an unsaturation and carbonyl group. IR spectrum indicates the presence of C-H str of C=C, C-H str of CH3, C-H str of CH, C=O, C=C bend, C-N, C-H bend, C-O aromatic, C-O aliphatic groups. NMR spectroscopy also suggests the existence of aliphatic and aromatic carbons and CHO group. Considering the UV, IR, NMR and LCMS spectroscopic details, the structure of the compound obtained is shown above.
References
|
||||||||||||||||||||||||||||||||||||